 The secondary endpoints are: EQ-5D score, activities of daily living (ADL), bony union, upper cervical stability and mortality. At 12 months, a dynamic radiographical investigation of upper cervical stability is performed. All participants are monitored with regard to the NDI, EuroQol score (EQ-5D), socio-demographics and computed tomography (CT) at the time of injury, at 6 weeks, 3 months and 12 months. The surgical group is treated with a posterior C1–C2 fusion. The non-surgical group is fitted with a rigid cervical collar for 12 weeks. By considering a 1-year mortality forecast of 29%, up to 25 participants are recruited in each group. A minimum of 16 patients are needed in each group to test the superiority with 80% power. The minimal clinically important difference of the NDI is 3.5 points. Excluded are patients with an American Society of Anaesthesiologists (ASA) score ≥ 4, dementia nursing care or anatomical cervical anomalies. 
Fifty consecutive patients aged ≥ 75 years, with displaced type-2 odontoid fracture, are randomised to non-surgical or surgical treatment. The Uppsala Study on Odontoid Fracture Treatment (USOFT) is a multicentre, open-label, randomised controlled superiority trial evaluating the clinical superiority of the surgical treatment of type-2 odontoid fractures, with a 1-year Neck Disability Index (NDI) as the primary endpoint. Due to the paucity of evidence, the treatment decision is often left to the discretion of the expert surgeon. 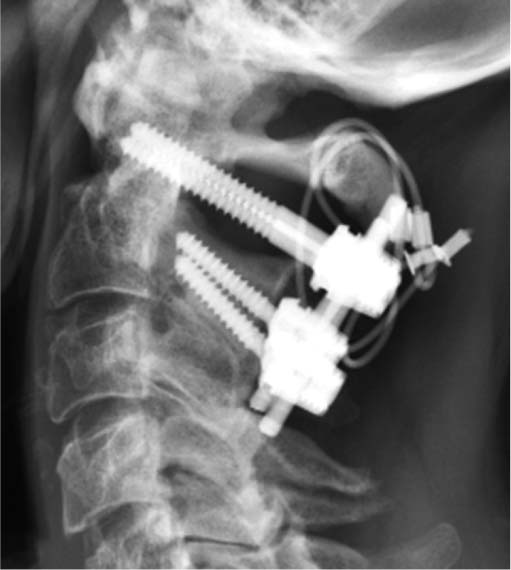
Displaced odontoid fractures in the elderly are treated non-surgically with a cervical collar or surgically with C1–C2 fusion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
